A group of bacteria called TM7 live in the human body by growing on the surfaces of other microbes, known as host bacteria. Ever since this arrangement was discovered, many scientists have assumed TM7 were harmful to humans, while their host bacteria were health promoting. Studies have shown higher abundance of TM7 in diseases including vaginosis, inflammatory bowel disease, and periodontitis, and lower abundance of host bacteria, further suggesting TM7 are pathogenic. But a new paper published today by Forsyth Institute researchers and their colleagues finds the opposite effect—TM7 decreased periodontal inflammation and bone loss in a mouse model.
The paper, Episymbiotic Saccharibacteria suppresses gingival inflammation and bone loss in mice through host bacterial modulation, was published online today in the journal Cell Host & Microbe. It describes the first time TM7 has been systematically cultured from human periodontal disease samples, isolated from their host bacteria, and studied in an animal model.
Dr. Batbileg Bor, Assistant Member of Staff at The Forsyth Institute and corresponding author of the paper, said he and his team took plaque samples from patients with gum disease and isolated multiple species of TM7 bacteria from the samples. To understand how TM7 impacts bone loss and inflammation, they devised an experiment with three groups of mice. In one group, a clean ligature was placed around one of the mouse’s molars, establishing a baseline. In the second group, the ligature was soaked in TM7’s host bacteria alone and then wrapped around the molar. In the third group, researchers soaked the ligature in TM7’s host bacteria, plus TM7 bacteria from the periodontal samples, and then placed it on the molar.
“Our findings were the opposite of what we and others expected,” Bor said. The mice fitted with the ligatures soaked in TM7’s host bacteria alone experienced more intense bone loss and inflammation than the established baseline, while the mice that got ligatures with TM7 experienced a decrease in bone loss and inflammation.
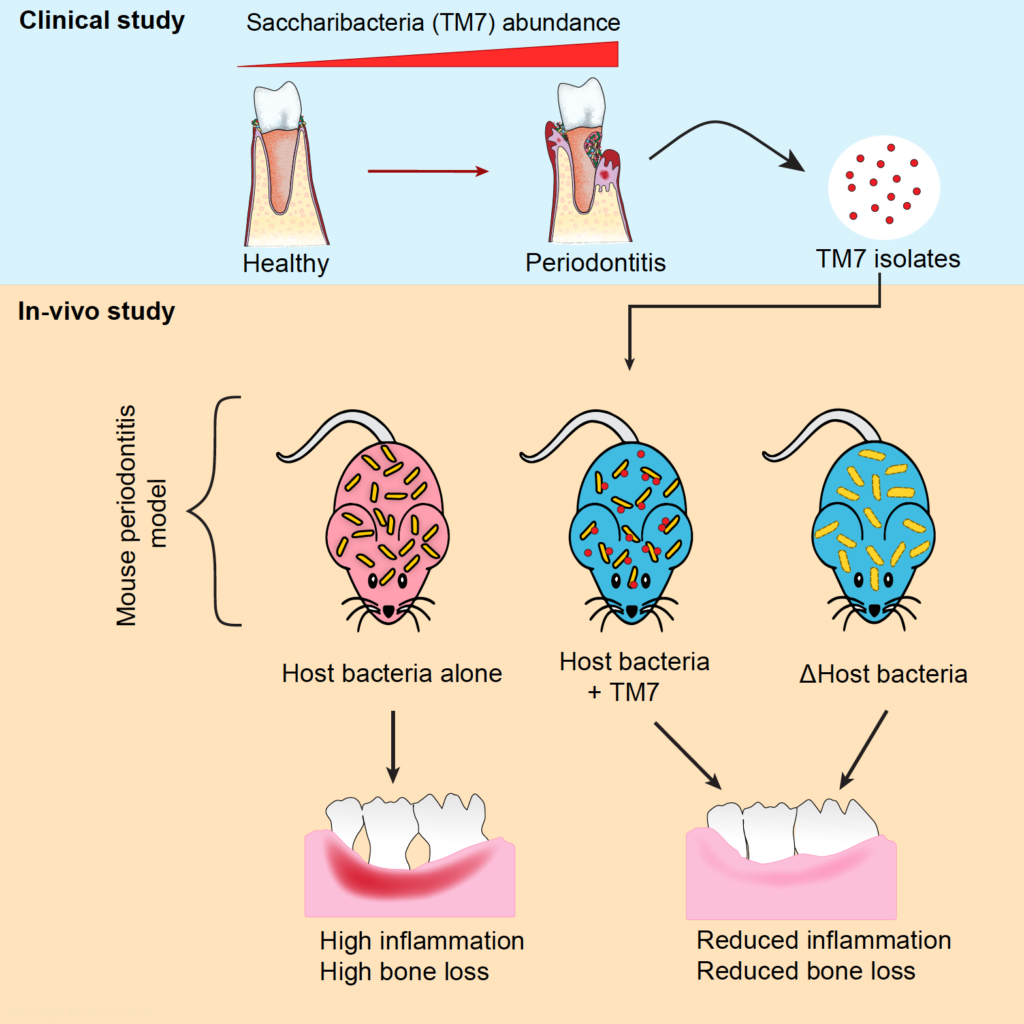
This finding turns upside down the idea that TM7 are pathogenic and their host bacteria are health associated. In fact, Bor says he now believes TM7 may be protective against disease, while their host bacteria are the harmful ones.
Co-authors of the paper include Postdoctoral Research Fellow Deepak Chouhan and Research Associate Otari Chipashvili from Forsyth, as well as Daniel Utter, a Postdoctoral Researcher at California Institute of Technology.
The researchers discovered two important mechanisms TM7 are modulating in the mouse model to reduce inflammation and bone loss, both by altering the behavior of their host bacteria. The first is decreased production of a collagen binding protein in the host bacteria, where collagen is commonly found in the network of molecules that form tissues. The second is disruption of the host bacterial metabolism in the inflammatory environment, which TM7 accomplish by shutting down the genes that break down sialic acid, one of the most common carbohydrates that coats the surface of many human cells.
“From these findings, we are debunking the previous hypothesis that TM7 are pathogenic and bad for you while the host bacteria are good and health associated. We think it might actually be the other way around,” Bor said.
TM7 are ultrasmall bacteria with tiny genomes that need to live off other organisms to survive. They are found in humans and other mammals, soil, water, and many other varied environments. There are dozens of different species within the TM7 phyla, and TM7 is one of 73 different phyla under the Candidate Phyla Radiation lineage. Most of the bacteria within this group have yet to be cultured in the lab and therefore remain a mystery to scientists, despite representing more than 25 percent of all bacterial diversity on Earth.
Previous studies have found TM7 in the dental tartar of human ancestors, suggesting these bacteria have been coevolving with humans for thousands of years.

“You can infer that TM7 are part of your extended immune system. It’s like a protective part of you that is just hanging out in your mouth, and when there is inflammation, it can try to reduce that,” Chipashvili explained.
While this research is still in early stages, Bor hopes to ultimately harness TM7 to promote better oral health. One potential application would be similar to phage therapy, or the use of bacterial viruses to kill pathogenic bacteria without needing drugs or antibiotics.
“If our hypothesis is proven right, TM7 could function in the same way, but there is a lot that needs to be done before we can get to that stage,” Utter said. “Culturing these bacteria and understanding their genome was the first major step. This new study marks another major step as the first in vivo experimental study to upend previous notions of TM7’s role in health and disease of oral microbial communities. We are looking forward to what may come out of these studies.”
Learn more about the Bor Lab.
